Vascular Health as a Root Driver for Cognitive Health
Protocol development in integrative medicine is not typically a simple process. Individuals require individualized care, and what works for one patient may not work for another.
To establish these protocols, we first developed a Rating Scale that could be used to discern the rigor of evidence supporting a specific nutrient’s therapeutic effect.
The following protocols were developed using only A through D-quality evidence.
Introduction
What Is Cognitive Support for Healthy Aging?
Cognitive health encompasses the capacity to maintain attention, memory, and mental clarity by supporting the systems that influence brain function. These include mitochondrial efficiency, antioxidant capacity, inflammation balance, adequate nutrient status, and circadian rhythm alignment. (Jost 2025)(Melzer 2021)(Wright 2015)
This clinical guide is designed to act as a structured, evidence-informed framework to support vascular-mediated mechanisms known to affect brain health throughout the lifespan: endothelial function, inflammation, oxidative stress, and lipid metabolism.
Why Vascular Integrity Matters
The brain’s high metabolic demand makes it heavily reliant on an intricate vascular network to deliver a steady supply of oxygen and nutrients to its tissues. Even modest disruptions in cerebral blood flow can disrupt neuronal activity and energy metabolism. The cerebral microvasculature is particularly vulnerable to endothelial dysfunction, arterial stiffness, and impaired nitric oxide (NO) signaling, which can lead to cerebral hypoperfusion and contribute to neurodegeneration and cognitive decline. (Ashby 2021)(Claassen 2021)(Liu 2021)
Cerebrovascular disease contributes to dementia in up to 75% of cases, and vascular cognitive impairment and dementia (VICD) is the second most common cause of dementia after Alzheimer’s disease (AD). (Sachdev 2026) Premature cardiovascular disease (CVD) predicts lower cognition and greater white matter injury over time. (Jiang 2023)
Purpose of the Clinical Guide
The Vascular Health as a Root Driver for Cognitive Health clinical guide was designed to:
-
- Simplify decision-making using standardized, evidence-rated nutrient interventions.
- Integrate laboratory and biomarker data to identify modifiable vascular contributors to cognitive health.
- Complement the Cognitive Essentials for Healthy Aging clinical guide, enabling providers to integrate vascular-focused strategies alongside foundational interventions.
Essential Labs
Endothelial Function Markers
Endothelial dysfunction is a pathological state in which the endothelium loses its normal regulatory functions. It is characterized by reduced NO bioavailability and increased vascular oxidative stress, inflammation, and vasoconstrictive signaling. (Hadi 2005) Peripheral endothelial dysfunction independently predicts future cognitive and cerebrovascular events. (Guse 2025)
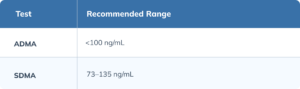
Asymmetric Dimethylarginine (ADMA) and Symmetric Dimethylarginine (SDMA)
ADMA and SDMA are methylated derivatives of L-arginine produced during normal cellular protein metabolism. They are both inhibitors of nitric oxide synthase (NOS), the enzyme responsible for producing NO. (Tsikas 2018) ADMA- and SDMA-mediated NOS inhibition promotes vasoconstriction, the progression of atherosclerosis, and impairs cerebral blood flow. (Asif 2013) A meta-analysis found that patients with dementia had significantly higher circulating concentrations of ADMA and SDMA compared to healthy controls. (Zinellu 2023)
Inflammation Markers
Through its effects on vascular integrity and neuroinflammation, inflammation represents a key mechanistic bridge between CVD and cognitive dysfunction. Chronic low-grade inflammation contributes to endothelial dysfunction, arterial stiffness, and atherosclerosis, thereby impairing cerebral blood flow and increasing the vulnerability of brain tissue to ischemic injury. (Rajeev 2023) Elevated inflammatory markers have consistently been linked to structural brain changes and reduced cognitive performance. (Han 2021)(Sartori 2012)
Fibrinogen
Fibrinogen is a protein involved in the clotting cascade, and elevated levels are associated with increased CV risk and neuroinflammation. (Patel 2024) In patients with MCI, high plasma fibrinogen has been correlated to poor attention and executive function regardless of other vascular risk factors. (Pyun 2020)

Oxidative Stress Markers
Oxidative stress refers to an imbalance between the production of reactive oxygen species (ROS) and the body’s ability to neutralize them with antioxidant defenses. (Pizzino 2017) Oxidative stress contributes to chronic cerebral hypoperfusion, which precedes cognitive decline and accelerates neurodegenerative processes via mitochondrial dysfunction, neuroinflammation, and neuronal apoptosis. (Liu 2012)(Lv 2025)
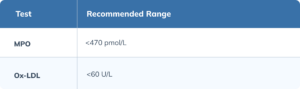
Myeloperoxidase (MPO)
MPO is a pro-oxidant enzyme released by activated immune cells during inflammatory responses. It catalyzes the formation of ROS that can drive lipid peroxidation and vascular injury, contributing to vascular dysfunction. (Khan 2018) Higher plasma MPO levels correlate with AD presence, disease severity, and amyloid burden. (Rivera Antonio 2025)(Tzikas 2014)
Oxidized Low-Density Lipoprotein (OxLDL)
OxLDL is low-density lipoprotein (LDL) that has undergone oxidative modification of its lipid and protein components. OxLDL promotes endothelial dysfunction, atherosclerotic plaque formation, and vascular inflammation, all of which compromise cerebral perfusion. (Jiang 2022)
Population studies have demonstrated that individuals with elevated OxLDL levels have higher rates of cognitive impairment compared to those with lower levels, with each standard deviation increase in OxLDL associated with up to a 26% higher risk of cognitive impairment. (Wang 2018)(Zhao 2025)
Basic Lipid Panel
[In-app] SKU: Cardio IQ Lipid Panel, QUS041-QD0098, and hyperlink
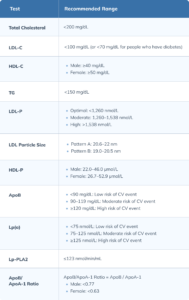
Total Cholesterol (TC)
High midlife TC is associated with over a two-fold increased risk of MCI and AD later in life. This association persists even after adjusting for vascular risk factors. (Anstey 2017)(Schilling 2017)
Compared to people with stable cholesterol, evidence also suggests that high fluctuations in TC among older adults are associated with a 60% higher risk of dementia and 23% increased likelihood of cognitive decline, potentially due to destabilization of atherosclerotic plaques that impairs cerebral blood flow. (American Heart Association 2024)
LDL Cholesterol (LDL-C)
Similar to TC, elevated midlife LDL-C (measured before age 65) is associated with increased dementia risk more than ten years later. (Iwagami 2021) Evidence suggests that LDL-C is a modifiable risk factor for AD, independent of apolipoprotein E (APOE) genotype, based on autopsy findings that correlated higher LDL-C levels with increased beta-amyloid plaques, neurofibrillary tangles, and cerebral amyloid angiopathy. (Wingo 2022)
High LDL-C also increases the risk of stroke, and 25–30% of survivors of ischemic stroke will develop vascular cognitive impairment or vascular dementia. (Kalaria 2016)
High-Density Lipoprotein (HDL) Cholesterol (HDL-C)
HDL-C is cardioprotective due to its role in reverse cholesterol transport, antioxidant activity, and anti-inflammatory effects that support endothelial function. Low HDL-C levels are associated with increased CVD risk and have also been linked to adverse brain outcomes, including reduced gray matter volume, which may contribute to an elevated risk of future cognitive decline. (Perswani 2024)(Ward 2010)
However, evidence also suggests that the relationship between HDL-C and health outcomes is not linear. Large observational studies demonstrate a U-shaped association between HDL-C levels and both CVD and dementia risk, such that individuals at the lowest and highest HDL-C percentiles have higher rates of atherosclerotic cardiovascular disease (ASCVD) mortality and dementia incidence compared with those in the mid-range. (Ferguson 2023)(Razavi 2023)
Triglycerides (TG)
Higher TG levels in midlife are associated with a 1.62-fold increased risk of non-Alzheimer’s dementia, a 1.25-fold increased risk of AD, and a 1.57-fold increased risk of ischemic stroke after multivariable adjustment. (Nordestgaard 2021)
Midlife hypertriglyceridemia is also independently associated with a greater likelihood of Alzheimer’s-related brain β-amyloid and tau pathology 20 years later in cognitively healthy individuals. (Nägga 2018) The TG/HDL-C ratio exhibits a particularly strong association with cognitive impairment, with elevated ratios being linked to poorer cognitive performance in older adults. (Huang 2026)
Advanced Lipid Panel
LDL Particle Number (LDL-P)
LDL-C reflects how much cholesterol is being carried within LDL particles, whereas LDL-P is the actual number of LDL particles circulating in the blood. Elevated LDL-P indicates a higher atherogenic particle burden and is therefore a sensitive marker of CVD risk. (El Harchaoui 2007) In people with discordant LDL-P and LDL-C levels, LDL-P is the better predictor of ASCVD risk. (Bays 2014)
LDL Particle Size
Research indicates that small dense LDL (sdLDL) particles are particularly prone to penetrating the arterial wall, where they undergo oxidative modification and trigger inflammatory processes that drive atherosclerosis. (Ivanova 2017)
Higher levels of sdLDL are associated with cognitive decline. For example, in the Atherosclerosis Risk in Communities (ARIC) study, each 10 mg/dL higher sdLDL-C level was associated with a faster decline in global cognitive scores and Digit Symbol Substitution Test performance over time. (Pokharel 2019)
HDL Particle Number (HDL-P)
HDL-P appears to be a superior predictor of ASCVD risk compared to HDL-C, likely reflecting its better correlation to HDL functionality. (Mackey 2012)(Mora 2013) In the Multi-Ethnic Study of Atherosclerosis (MESA) study, HDL-P maintained its inverse association with coronary heart disease after adjustment for HDL-C, LDL-P, LDL-C, and TG, whereas HDL-C lost statistical significance and even showed weakly positive hazard ratios in joint models. (Mackey 2012)
Emerging research suggests that high HDL-P correlates with improved cognitive function. In the Study of Women’s Health Across the Nation (SWAN) HDL Ancillary Study, higher total midlife HDL-P was associated with better subsequent immediate recall, delayed recall, and processing speed. (Qi 2025)
Apolipoprotein B (ApoB)
ApoB is a structural protein of atherogenic lipoproteins, making it a direct measure of the total number of circulating atherogenic particles. Higher ApoB levels are associated with higher ASCVD risk and accelerated cognitive decline. (De Oliveira-Gomes 2024)(Pokharel 2019)
Studies demonstrate that individuals with elevated ApoB are at a significantly higher risk of a greater decline in global cognitive scores, memory, and visuospatial function compared to those with lower levels, with a magnitude comparable to carrying one APOE ε4 allele. (Lutski 2019)(Pokharel 2019)
Lipoprotein (a) (Lp[a])
Lp(a) is an LDL-like particle with an additional apolipoprotein(a) (apo[a]) component. Lp(a) levels are largely genetically determined, and the apo(a) component is associated with substantially increased atherogenicity compared with standard LDL. (Alebna 2023) Elevated Lp(a) may contribute to cerebrovascular disease, cognitive decline, and increased dementia risk by promoting ischemia and chronic inflammation. (Li 2021)(Röhr 2020)
Lipoprotein-Associated Phospholipase A2 (Lp-PLA2)
Lp-PLA2 is a calcium-independent serine lipase bound to LDL particles, known for its pro-inflammatory and oxidative properties. (De Stefano 2019) Elevated circulating Lp-PLA2 levels have been linked to a higher risk of dementia, independent of CV and inflammatory factors. (van Oijen 2006) Notably, each standard deviation increase in Lp-PLA2 mass has been associated with an 11% greater risk of developing dementia. (Fitzpatrick 2014)
ApoB/Apolipoprotein A-1 (ApoA-1) Ratio
The ApoB/ApoA-1 ratio reflects the balance between atherogenic and antiatherogenic lipoproteins and is a superior predictor of CV risk compared to traditional cholesterol measures, such as LDL-C. It is calculated by dividing ApoB by ApoA-1, with values >0.6 indicating a higher risk for major adverse CV events and myocardial infarction. (Walldius 2021)
Higher ApoB/ApoA-1 ratios are observed in individuals with MCI compared to cognitively normal peers and predict future cognitive decline in those who are initially unimpaired. (Song 2012)
Ingredients
Coenzyme Q10 (CoQ10)
Dosing: 200 mg of ubiquinol daily for 2–12 months (Daei 2024)(García-Carpintero 2021)
Supporting evidence:
- Evidence suggests that plasma CoQ10 levels are significantly associated with overall cognitive functioning and executive function, even after controlling for physical activity and nutritional status. (Fernández-Portero 2023) Up to 73% of patients with dementia may have low CoQ10 status, which correlates to higher serum amyloid-β (Aβ) levels, reduced antioxidant capacity, and lower Mini-Mental State Examination (MMSE) scores. (Chang 2022)
- Animal models suggest that supplementation with CoQ10 may help reduce amyloid pathology, decrease oxidative stress markers, and improve cognitive performance. (Asadbegi 2023)(Dumont 2011)
- A one-year randomized controlled trials (RCT) in 69 patients with MCI taking ubiquinol (the reduced form of CoQ10) showed gender-dependent improvements in cerebral vasoreactivity and reduced inflammation, particularly in male patients who achieved higher plasma CoQ10 levels (≥5 μg/mL). (García-Carpintero 2021)
MonitumRSTM (Green Seaweed [Monostroma spp.])
Dosing: 100 mg per day for two weeks (Shimada 2021)
Supporting evidence:
- MonitumRS™ is a rhamnan sulfate (RS) extract from the green seaweed Monostroma nitidum. (Burke 2023) Research indicates that RS exhibits multiple bioactive properties that support CV health and function, including anticoagulant, antioxidant, anti-obesity, antidiabetic, anti-inflammatory, and hypolipidemic effects. (Hoang 2015)(Suzuki 2020)
- A six-month case series assessed the effects of daily supplementation with MonitumRS™, the main active ingredient in a proprietary blend, in six high-risk patients with carotid atherosclerosis who were unresponsive to prior interventions. Carotid intima-media thickness (CIMT) ultrasound showed a significant average reduction in total plaque burden by 5.55mm. A statistically significant reduction of mean maximum plaque thickness of 0.89 mm in the right carotid and 1.11 mm in the left was also measured. (Burke 2023)
- RS may help prevent the development of atherosclerotic plaques by protecting the endothelial glycocalyx (EGX) layer of the vascular endothelium. (Burke 2023) An in vitro study found that high glucose degraded the EGX, while MonitumRS™ effectively restored it—exceeding its pre-damaged state by up to 103.8% within just 24 hours. (State Key Laboratory of Bioengineering 2023)
- In a preclinical study, RS from Monostroma nitidum improved hyperglycemia-induced cognitive decline in mice that were pretreated with nicotinamide/streptozotocin, fed a high-fat diet, and administered maltodextrin, then dosed orally at 75–750 mg/kg for four months. RS significantly lowered blood glucose levels, reduced hippocampal neuronal atrophy, and suppressed brain tumor necrosis factor-α (TNF-α) expression and microglial activation. Behavioral tests of memory function showed improvement trends, but lacked statistical significance. These results suggest RS may modulate neurological and cognitive function via vascular pathways, warranting further human studies on its cognitive benefits. (Miwa 2025)
Black Garlic Extract
Dosing: 170–250 mg of extract, or four black garlic cloves (12 g), daily for 12 weeks (Serrano 2023)(Villaño 2023)
Supporting evidence:
- The authors of a small RCT concluded that a garlic extract-containing proprietary blend helped support healthy blood pressure levels by improving NO availability. Results showed that the 12 patients taking the supplement daily achieved a mean reduction in systolic blood pressure of 11 mmHg. Reductions in diastolic blood pressure were also observed in patients with hypertension. Salivary NO levels rose within two hours of taking the supplement and remained elevated for the duration of the study. (Houston 2023) This is relevant as elevated blood pressure is a risk factor for age-related cognitive decline. (Pacholko 2024)
- A single-center, controlled clinical trial demonstrated that daily consumption of black garlic may improve parameters associated with CV risk. After 12 weeks, endothelial adhesion molecules, which promote vascular inflammation and plaque formation, were reduced in patients with and without hypercholesterolemia. ApoA-1 levels also increased in both patient groups, suggesting improved reverse cholesterol transport and atheroprotective effects. (Villaño 2023)
- Results from an RCT with 58 healthy adults indicate that fermented garlic extract enhances vascular function by boosting NO bioavailability. This was evidenced by significant reductions in systolic and diastolic blood pressure, lowered systolic and diastolic flow velocities in the common carotid arteries, increased regional cerebral blood flow in frontal and parietal cortical regions, and elevated peripheral skin temperature. (Baik 2022)
Anthocyanins
Dosing: Dose varies depending on the extract
Supporting evidence:
-
- Anthocyanins counteract oxidative stress and inflammation (major contributors to endothelial dysfunction, CVD, and neurodegenerative disease) by reducing ROS formation through antioxidant activities. (Speer 2020)
- Studies have found that anthocyanin supplementation may help reduce inflammatory markers, including TNF-α, CRP, interleukin-6 (IL-6), and interleukin-1β (IL-1β). Anthocyanins may also enhance antioxidant capacity, as reflected by increased total radical-trapping antioxidant parameter (TRAP) and ferric reducing antioxidant power (FRAP) values. (Li 2015)(Xu 2021)(Zhu 2013)
- RCTs suggest that anthocyanins may protect cognitive function—including short-term memory, verbal learning, working memory, executive function, visual-spatial function, psychomotor skills, attention, and semantic memory— in part by enhancing vascular function and mitigating cardiovascular risk factors. (Ellis 2024)(Lorzadeh 2025)
Beetroot Extract
Dosing: 400 mg per day of nitrate (Miraftabi 2021)
Supporting evidence:
- Beetroot is a rich source of inorganic nitrate, which is reduced to nitrite by oral bacteria and then subsequently converted to NO. This NO-dependent pathway promotes vasodilation, thereby improving oxygen and nutrient delivery. (Dos Santos Baião 2021)(Premont 2020)
- Betanin, a phytochemical found in beetroot, may exert cardioprotective actions through direct free-radical scavenging, preservation of lipids and LDL particles, activation of the nuclear factor erythroid-2-related factor 2 (Nrf2) antioxidant pathway, and suppression of nuclear factor κ-B (NF-κB) inflammatory signaling. (Silva 2022) Recent evidence also suggests that these anti-inflammatory and antioxidant mechanisms confer multi-target neuroprotective actions in models of AD, Parkinson’s disease, ischemia-reperfusion injury, and other forms of neurodegeneration. (Ştefănescu 2025)
- A systematic review of 43 RCTs concluded that supplemental inorganic nitrate derived from beetroot is associated with significant improvements in endothelial function, reflected by a 0.62% increase in flow-mediated dilation, and reduced arterial stiffness, measured by a 0.27 m/s decrease in pulse wave velocity. (Bahrami 2021)
- In a double-blind crossover trial of 44 healthy adults, a single dose of nitrate-standardized beetroot extract administered 90 minutes prior to neuropsychological testing significantly enhanced memory consolidation, frontal lobe-related executive functions, and cognitive flexibility compared to the placebo. (Vaccaro 2023)
Anthocyanins
Dosing: Dose varies depending on the extract
Supporting evidence:
-
- Anthocyanins counteract oxidative stress and inflammation (major contributors to endothelial dysfunction, CVD, and neurodegenerative disease) by reducing ROS formation through antioxidant activities. (Speer 2020)
- Studies have found that anthocyanin supplementation may help reduce inflammatory markers, including TNF-α, CRP, interleukin-6 (IL-6), and interleukin-1β (IL-1β). Anthocyanins may also enhance antioxidant capacity, as reflected by increased total radical-trapping antioxidant parameter (TRAP) and ferric reducing antioxidant power (FRAP) values. (Li 2015)(Xu 2021)(Zhu 2013)
- RCTs suggest that anthocyanins may protect cognitive function—including short-term memory, verbal learning, working memory, executive function, visual-spatial function, psychomotor skills, attention, and semantic memory— in part by enhancing vascular function and mitigating cardiovascular risk factors. (Ellis 2024)(Lorzadeh 2025)
- Blackcurrant extract
-
-
- In humans, clinical trials suggest that blackcurrant supplementation may support prefrontal cerebral hemodynamics and improve aspects of working memory, subjective alertness, and mental fatigue. (Lomiwes 2024)(Nilsson 2017)(Watson 2026)
- In rodents, supplementation with anthocyanin-rich blackcurrant has been shown to help enhance spatial learning and memory and prevent scopolamine-induced deficits by normalizing cholinesterase activity and reducing oxidative stress. (da Costa 2024)(Lomiwes 2025)
-
- Bilberry extract
-
- A four-week, randomized, double-blind, placebo-controlled pilot trial in adults using 160 mg/day of standardized bilberry extract found that supplementation was associated with significant reductions in tension, depression, and confusion scores compared with placebo. Trends toward improvements in cognitive flexibility and executive functioning were also observed, although these outcomes did not reach statistical significance. (Kara 2025)
- In vitro and preclinical evidence suggest that anthocyanin-rich bilberry extracts exert neuroprotective effects, including antioxidant activity, attenuation of Aβ- and ROS-induced toxicity, increased expression of brain-derived neurotrophic factor (BDNF), and modulation of neurotransmitter targets that are mechanistically relevant to mood and cognition. (Choi 2014)(Kara 2025)(König 2024)
Disclaimer
The Fullscript Integrative Medical Advisory team has developed or collected these protocols from practitioners and supplier partners to help health care practitioners make decisions when building treatment plans. By adding this protocol to your Fullscript template library, you understand and accept that the recommendations in the protocol are for initial guidance and may not be appropriate for every patient.
References
- Alebna, P. L., & Mehta, A. (2023, September 19). An update on lipoprotein(a): The latest on testing, treatment, and guideline recommendations. American College of Cardiology. https://www.acc.org/Latest-in-Cardiology/Articles/2023/09/19/10/54/An-Update-on-Lipoprotein-a
- American Heart Association. (2024, November 11). Unexplained changes in cholesterol may help identify older adults at risk for dementia. American Heart Association. https://newsroom.heart.org/news/unexplained-changes-in-cholesterol-may-help-identify-older-adults-at-risk-for-dementia
- Anstey, K. J., Ashby-Mitchell, K., & Peters, R. (2017). Updating the evidence on the association between serum cholesterol and risk of late-life dementia: Review and meta-analysis. Journal of Alzheimer’s Disease, 56(1), 215–228. https://doi.org/10.3233/jad-160826
- Asadbegi, M., Komaki, H., Faraji, N., Taheri, M., Safari, S., Raoufi, S., Kourosh-Arami, M., Golipoor, Z., & Komaki, A. (2023). Effectiveness of coenzyme Q10 on learning and memory and synaptic plasticity impairment in an aged Aβ-induced rat model of Alzheimer’s disease: A behavioral, biochemical, and electrophysiological study. Psychopharmacology, 240(4), 951–967. https://doi.org/10.1007/s00213-023-06338-2
- Ashby, J. W., & Mack, J. J. (2021). Endothelial control of cerebral blood flow. The American Journal of Pathology, 191(11), 1906–1916. https://doi.org/10.1016/j.ajpath.2021.02.023
- Asif, M., Soiza, R. L., McEvoy, M., & Mangoni, A. A. (2013). Asymmetric dimethylarginine: A possible link between vascular disease and dementia. Current Alzheimer Research, 10(4), 347–356. https://doi.org/10.2174/1567205011310040001
- Bahrami, L. S., Arabi, S. M., Feizy, Z., & Rezvani, R. (2021). The effect of beetroot inorganic nitrate supplementation on cardiovascular risk factors: A systematic review and meta-regression of randomized controlled trials. Nitric Oxide, 115, 8–22. https://doi.org/10.1016/j.niox.2021.06.002
- Baik, J. S., Min, J. H., Ju, S. M., Ahn, J. H., Ko, S. H., Chon, H. S., Kim, M. S., & Shin, Y. I. (2022). Effects of fermented garlic extract containing nitric oxide metabolites on blood flow in healthy participants: A randomized controlled trial. Nutrients, 14(24), 5238. https://doi.org/10.3390/nu14245238
- Bays, H. E., Jones, P. H., Brown, W. V., & Jacobson, T. A. (2014). National Lipid Association annual summary of clinical lipidology 2015. Journal of Clinical Lipidology, 8(6), S1–S36. https://doi.org/10.1016/j.jacl.2014.10.002
- Burke, K. L., & Jennings, I. G. (2023). The effect of a dietary supplement containing rhamnan sulfate from Monostroma nitidum on carotid atherosclerotic plaque: A case series. Integrative Medicine (Encinitas), 22(6), 30–37. https://pubmed.ncbi.nlm.nih.gov/38404609/
- Chang, P.-S., Chou, H.-H., Lai, T.-J., Yen, C.-H., Pan, J.-C., & Lin, P.-T. (2022). Investigation of coenzyme Q10 status, serum amyloid-β, and tau protein in patients with dementia. Frontiers in Aging Neuroscience, 14, 910289. https://doi.org/10.3389/fnagi.2022.910289
- Choi, Y.-H., Kwon, H.-S., Shin, S.-G., & Chung, C.-K. (2014). Vaccinium uliginosum L. improves amyloid β protein-induced learning and memory impairment in Alzheimer’s disease in mice. Preventive Nutrition and Food Science, 19(4), 343–347. https://doi.org/10.3746/pnf.2014.19.4.343
- Claassen, J. A. H. R., Thijssen, D. H. J., Panerai, R. B., & Faraci, F. M. (2021). Regulation of cerebral blood flow in humans: Physiology and clinical implications of autoregulation. Physiological Reviews, 101(4), 1487–1559. https://doi.org/10.1152/physrev.00022.2020
- da Costa, P., Schetinger, M. R. C., Baldissarelli, J., Stefanello, N., Lopes, T. F., Reichert, K. P., Assmann, C. E., Bottari, N. B., Miron, V. V., Vargas, F. F. A., Gutierres, J. M., da Cruz, I. B. M., & Morsch, V. M. (2024). Blackcurrant (Ribes nigrum L.) improves cholinergic signaling and protects against chronic Scopolamine-induced memory impairment in mice. Journal of Psychopharmacology, 38(12), 1170–1183. https://doi.org/10.1177/02698811241273776
- Daei, S., Ildarabadi, A., Goodarzi, S., & Mohamadi-Sartang, M. (2024). Effect of coenzyme Q10 supplementation on vascular endothelial function: A systematic review and meta-analysis of randomized controlled trials. High Blood Pressure & Cardiovascular Prevention, 31(2), 113–126. https://doi.org/10.1007/s40292-024-00630-8
- De Oliveira-Gomes, D., Joshi, P. H., Peterson, E. D., Rohatgi, A., Khera, A., & Navar, A. M. (2024). Apolipoprotein B: Bridging the gap between evidence and clinical practice. Circulation, 150(1), 62–79. https://doi.org/10.1161/circulationaha.124.068885
- De Stefano, A., Mannucci, L., Tamburi, F., Cardillo, C., Schinzari, F., Rovella, V., Nisticò, S., Bennardo, L., Di Daniele, N., & Tesauro, M. (2019). Lp-PLA2, a new biomarker of vascular disorders in metabolic diseases. International Journal of Immunopathology and Pharmacology, 33, 2058738419827154. https://doi.org/10.1177/2058738419827154
- Dos Santos Baião, D., Vieira Teixeira da Silva, D., & Margaret Flosi Paschoalin, V. (2021). A narrative review on dietary strategies to provide nitric oxide as a non-drug cardiovascular disease therapy: Beetroot formulations – A smart nutritional intervention. Foods, 10(4), 859. https://doi.org/10.3390/foods10040859
- Dumont, M., Kipiani, K., Yu, F., Wille, E., Katz, M., Calingasan, N. Y., Gouras, G. K., Lin, M. T., & Beal, M. F. (2011). Coenzyme Q10 decreases amyloid pathology and improves behavior in a transgenic mouse model of Alzheimer’s disease. Journal of Alzheimer’s Disease, 27(1), 211–223. https://doi.org/10.3233/jad-2011-110209
- El Harchaoui, K., van der Steeg, W. A., Stroes, E. S. G., Kuivenhoven, J. A., Otvos, J. D., Wareham, N. J., Hutten, B. A., Kastelein, J. J. P., Khaw, K.-T., & Boekholdt, S. M. (2007). Value of low-density lipoprotein particle number and size as predictors of coronary artery disease in apparently healthy men and women: The EPIC-Norfolk Prospective Population Study. Journal of the American College of Cardiology, 49(5), 547–553. https://doi.org/10.1016/j.jacc.2006.09.043
- Ellis, L. R., Boesch, C., & Dye, L. (2024). Effects of anthocyanins on cognition and vascular function: A systematic review. Molecular Nutrition & Food Research, 68(13), e2300502. https://doi.org/10.1002/mnfr.202300502
- Ferguson, E. L., Zimmerman, S. C., Jiang, C., Choi, M., Swinnerton, K., Choudhary, V., Meyers, T. J., Hoffmann, T. J., Gilsanz, P., Oni-Orisan, A., Whitmer, R. A., Risch, N., Krauss, R., Schaefer, C. A., & Glymour, M. M. (2023). Low- and high-density lipoprotein cholesterol and dementia risk over 17 years of follow-up among members of a large health care plan. Neurology, 101(21), e2172–e2184. https://doi.org/10.1212/WNL.0000000000207876
- Fernández-Portero, C., Amián, J. G., Bella, R., López-Lluch, G., & Alarcón, D. (2023). Coenzyme Q10 levels associated with cognitive functioning and executive function in older adults. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 78(1), 1–8. https://doi.org/10.1093/gerona/glac152
- Fitzpatrick, A. L., Irizarry, M. C., Cushman, M., Jenny, N. S., Chi, G. C., & Koro, C. (2014). Lipoprotein-associated phospholipase A2 and risk of dementia in the Cardiovascular Health Study. Atherosclerosis, 235(2), 384–391. https://doi.org/10.1016/j.atherosclerosis.2014.04.032
- Frydrych, A., Kulita, K., Jurowski, K., & Piekoszewski, W. (2025). Lipids in clinical nutrition and health: Narrative review and dietary recommendations. Foods, 14(3), 473. https://doi.org/10.3390/foods14030473
- García-Carpintero, S., Domínguez-Bértalo, J., Pedrero-Prieto, C., Frontiñán-Rubio, J., Amo-Salas, M., Durán-Prado, M., García-Pérez, E., Vaamonde, J., & Alcain, F. J. (2021). Ubiquinol supplementation improves gender-dependent cerebral vasoreactivity and ameliorates chronic inflammation and endothelial dysfunction in patients with mild cognitive impairment. Antioxidants, 10(2), 143. https://doi.org/10.3390/antiox10020143
- Guse, E.-C., Cotet, I.-G., Mateescu, D.-M., Muresan, C.-O., Gavrilescu, D.-M., Marginean, A., Toma, A.-O., Ilie, A.-C., Halas, R., Badalica-Petrescu, M., & Bredicean, A.-C. (2025). Peripheral microvascular and endothelial dysfunction as predictors of cognitive decline and small vessel disease: A systematic review and meta-analysis. Journal of Clinical Medicine, 14(23), 8543. https://doi.org/10.3390/jcm14238543
- Hadi, H. A., Carr, C. S., & Al Suwaidi, J. (2005). Endothelial dysfunction: Cardiovascular risk factors, therapy, and outcome. Vascular Health and Risk Management, 1(3), 183–198. https://pmc.ncbi.nlm.nih.gov/articles/PMC1993955/
- Han, K.-M., & Ham, B.-J. (2021). How inflammation affects the brain in depression: A review of functional and structural MRI studies. Journal of Clinical Neurology, 17(4), 503–515. https://doi.org/10.3988/jcn.2021.17.4.503
- Hoang, M. H., Kim, J.-Y., Lee, J. H., You, S., & Lee, S.-J. (2015). Antioxidative, hypolipidemic, and anti-inflammatory activities of sulfated polysaccharides from Monostroma nitidum. Food Science and Biotechnology, 24(1), 199–205. https://doi.org/10.1007/s10068-015-0027-x
- Houston, M., Chen, C., D’Adamo, C. R., Papathanassiu, A. E., & Green, S. J. (2023). Effects of S-allylcysteine-rich garlic extract and dietary inorganic nitrate formula on blood pressure and salivary nitric oxide: An open-label clinical trial among hypertensive subjects. Cureus, 15(9), e45369. https://doi.org/10.7759/cureus.45369
- Huang, Y., Zheng, L., & Lyu, M.-M. (2026). Association between ratio of triglycerides to HDL-C and cognitive impairment: A longitudinal population-based analysis and Mendelian randomization study. The American Journal of Geriatric Psychiatry, 34(1), 1–14. https://doi.org/10.1016/j.jagp.2025.08.007
- Ivanova, E. A., Myasoedova, V. A., Melnichenko, A. A., Grechko, A. V., & Orekhov, A. N. (2017). Small dense low-density lipoprotein as biomarker for atherosclerotic diseases. Oxidative Medicine and Cellular Longevity, 2017, 1273042. https://doi.org/10.1155/2017/1273042
- Iwagami, M., Qizilbash, N., Gregson, J., Douglas, I., Johnson, M., Pearce, N., Evans, S., & Pocock, S. (2021). Blood cholesterol and risk of dementia in more than 1·8 million people over two decades: A retrospective cohort study. The Lancet Healthy Longevity, 2(8), e498–e506. https://doi.org/10.1016/s2666-7568(21)00150-1
- Jiang, H., Zhou, Y., Nabavi, S. M., Sahebkar, A., Little, P. J., Xu, S., Weng, J., & Ge, J. (2022). Mechanisms of oxidized LDL-mediated endothelial dysfunction and its consequences for the development of atherosclerosis. Frontiers in Cardiovascular Medicine, 9, 925923. https://doi.org/10.3389/fcvm.2022.925923
- Jiang, X., Lewis, C. E., Allen, N. B., Sidney, S., & Yaffe, K. (2023). Premature cardiovascular disease and brain health in midlife: The CARDIA Study. Neurology, 100(14), e1454–e1463. https://doi.org/10.1212/WNL.0000000000206825
- Jost, Z., & Kujach, S. (2025). Understanding cognitive decline in aging: Mechanisms and mitigation strategies – A narrative review. Clinical Interventions in Aging, 20, 459–469. https://doi.org/10.2147/cia.s510670
- Kalaria, R. N., Akinyemi, R., & Ihara, M. (2016). Stroke injury, cognitive impairment and vascular dementia. Biochimica et Biophysica Acta, 1862(5), 915–925. https://doi.org/10.1016/j.bbadis.2016.01.015
- Kara, M., Hasbal-Celikok, G., Wilson, J., Gómez-Serranillos, P., Yilmaz-Ozden, T., Öztas, E., Özhan, G., Sultan Zengin, Ö., Gómez-Serranillos, M. S., Owsianik, C., Rabbani, F., Tunç, M., Arda, N., Memon, N. M., Ujjan, I., Hawkins, K., Davis, J., Wilson, G., Petrangolini, G., & Khan, A. (2025). In vitro mechanistic studies and potential health benefits of a standardized bilberry extract in low mood and cognitive enhancement. Frontiers in Nutrition, 12, 1630147. https://doi.org/10.3389/fnut.2025.1630147
- Khan, A. A., Alsahli, M. A., & Rahmani, A. H. (2018). Myeloperoxidase as an active disease biomarker: Recent biochemical and pathological perspectives. Medical Sciences, 6(2), 33. https://doi.org/10.3390/medsci6020033
- König, S., Bakuradze, T., Jesser, S., Sreeja, H. A., Carlsson, M. J., Fahrer, J., Kins, S., & Richling, E. (2024). Influence of bilberry extract on neuronal cell toxicity. Biology, 13(6), 376. https://doi.org/10.3390/biology13060376
- Li, D., Zhang, Y., Liu, Y., Sun, R., & Xia, M. (2015). Purified anthocyanin supplementation reduces dyslipidemia, enhances antioxidant capacity, and prevents insulin resistance in diabetic patients. The Journal of Nutrition, 145(4), 742–748. https://doi.org/10.3945/jn.114.205674
- Li, J., Li, S., Pan, Y., Wang, M., Meng, X., Wang, Y., Zhao, X., & Wang, Y. (2021). Relationship between lipoprotein (a) [Lp(a)] and cognition in different ischemic stroke subtypes. Frontiers in Neurology, 12, 736365. https://doi.org/10.3389/fneur.2021.736365
- Liu, H., & Zhang, J. (2012). Cerebral hypoperfusion and cognitive impairment: The pathogenic role of vascular oxidative stress. International Journal of Neuroscience, 122(9), 494–499. https://doi.org/10.3109/00207454.2012.686543
- Liu, W., Chen, Z., Ortega, D., Liu, X., Huang, X., Wang, L., Chen, L., Sun, J., Hatsukami, T. S., Yuan, C., Li, H., & Yang, J. (2021). Arterial elasticity, endothelial function and intracranial vascular health: A multimodal MRI study. Journal of Cerebral Blood Flow & Metabolism, 41(6), 1390–1397. https://doi.org/10.1177/0271678×20956950
- Lomiwes, D., Günther, C. S., Bloor, S. J., Trower, T. M., Ngametua, N., Kanon, A. P., Jensen, D. A., Lo, K., Sawyer, G., Walker, E. G., Hedderley, D., & Cooney, J. M. (2024). Identification of sarmentosin as a key bioactive from blackcurrants (Ribes nigrum) for inhibiting platelet monoamine oxidase in humans. Journal of Agricultural and Food Chemistry, 72(30), 16777–16789. https://doi.org/10.1021/acs.jafc.4c03802
- Lomiwes, D., Kanon, A. P., Ha, B., Cooney, J. M., Scholey, A., Lyall, K. A., Jensen, D. J., & Hurst, R. D. (2025). Short-term New Zealand “Blackadder” blackcurrant juice supplementation improves learning and memory in young adult rats. International Journal of Molecular Sciences, 26(23), 11568. https://doi.org/10.3390/ijms262311568
- Lorzadeh, E., Weston-Green, K., Roodenrys, S., Rosario, V. do, Kent, K., & Charlton, K. (2025). The effect of anthocyanins on cognition: A systematic review and meta-analysis of randomized clinical trial studies in cognitively impaired and healthy adults. Current Nutrition Reports, 14(1), 23. https://doi.org/10.1007/s13668-024-00595-z
- Lutski, M., Weinstein, G., Goldbourt, U., & Tanne, D. (2019). Plasma lipids, apolipoproteins, and subsequent cognitive decline in men with coronary heart disease. Journal of Alzheimer’s Disease, 67(3), 827–837. https://doi.org/10.3233/JAD-180849
- Lv, H., Liu, Y., Yang, X., Xu, X., Zhou, J., & Yu, W. (2025). Research progress on the role of oxidative stress in the pathogenesis of vascular dementia and its treatment. Journal of Stroke and Cerebrovascular Diseases, 34(12), 108475. https://doi.org/10.1016/j.jstrokecerebrovasdis.2025.108475
- Mackey, R. H., Greenland, P., Goff, D. C., Lloyd-Jones, D. M., Sibley, C. T., & Mora, S. (2012). High-density lipoprotein cholesterol and particle concentrations, carotid atherosclerosis, and coronary events: MESA (multi-ethnic study of atherosclerosis). Journal of the American College of Cardiology, 60(6), 508–516. https://doi.org/10.1016/j.jacc.2012.03.060
- Melzer, T. M., Manosso, L. M., Yau, S., Gil-Mohapel, J., & Brocardo, P. S. (2021). In pursuit of healthy aging: Effects of nutrition on brain function. International Journal of Molecular Sciences, 22(9), 5026. https://doi.org/10.3390/ijms22095026
- Miraftabi, H., Avazpoor, Z., Berjisian, E., Sarshin, A., Rezaei, S., Domínguez, R., Reale, R., Franchini, E., Samanipour, M. H., Koozehchian, M., Willems, M., Rafiei, R., & Naderi, A. (2021). Effects of beetroot juice supplementation on cognitive function, aerobic and anaerobic performances of trained male taekwondo athletes: A pilot study. International Journal of Environmental Research and Public Health, 18(19), 10202. https://doi.org/10.3390/ijerph181910202
- Miwa, T., Sato, M., Ma, N., Hiramoto, K., Terasawa, M., & Suzuki, K. (2025). Rhamnan sulfate from the seaweed Monostroma nitidum may improve cognitive impairment caused by hyperglycemia. Marine Drugs, 23(6), 250. https://doi.org/10.3390/md23060250
- Mora, S., Glynn, R. J., & Ridker, P. M. (2013). High-density lipoprotein cholesterol, size, particle number, and residual vascular risk after potent statin therapy. Circulation, 128(11), 1189–1197. https://doi.org/10.1161/circulationaha.113.002671
- Nägga, K., Gustavsson, A.-M., Stomrud, E., Lindqvist, D., van Westen, D., Blennow, K., Zetterberg, H., Melander, O., & Hansson, O. (2018). Increased midlife triglycerides predict brain β-amyloid and tau pathology 20 years later. Neurology, 90(1), e73–e81. https://doi.org/10.1212/wnl.0000000000004749
- Nilsson, A., Salo, I., Plaza, M., & Björck, I. (2017). Effects of a mixed berry beverage on cognitive functions and cardiometabolic risk markers; A randomized cross-over study in healthy older adults. PLoS One, 12(11), e0188173. https://doi.org/10.1371/journal.pone.0188173
- Nordestgaard, L. T., Christoffersen, M., Afzal, S., Nordestgaard, B. G., Tybjærg-Hansen, A., & Frikke-Schmidt, R. (2021). Triglycerides as a shared risk factor between dementia and atherosclerotic cardiovascular disease: A study of 125727 individuals. Clinical Chemistry, 67(1), 245–255. https://doi.org/10.1093/clinchem/hvaa269
- Pacholko, A., & Iadecola, C. (2024). Hypertension, neurodegeneration, and cognitive decline. Hypertension, 81(5), 991–1007. https://doi.org/10.1161/hypertensionaha.123.21356
- Patel, S., Govindarajan, V., Chakravarty, S., & Dubey, N. (2024). From blood to brain: Exploring the role of fibrinogen in the pathophysiology of depression and other neurological disorders. International Immunopharmacology, 143(Pt 1), 113326. https://doi.org/10.1016/j.intimp.2024.113326
- Perswani, P., Ismail, S. M., Mumtaz, H., Uddin, N., Asfand, M., Khalil, A. B. B., Ijlal, A., Khan, S. E., Usman, M., Younas, H., & Rai, A. (2024). Rethinking HDL-C: An in-depth narrative review of its role in cardiovascular health. Current Problems in Cardiology, 49(2), 102152. https://doi.org/10.1016/j.cpcardiol.2023.102152
- Pizzino, G., Irrera, N., Cucinotta, M., Pallio, G., Mannino, F., Arcoraci, V., Squadrito, F., Altavilla, D., & Bitto, A. (2017). Oxidative stress: Harms and benefits for human health. Oxidative Medicine and Cellular Longevity, 2017, 8416763. https://doi.org/10.1155/2017/8416763
- Pokharel, Y., Mouhanna, F., Nambi, V., Virani, S. S., Hoogeveen, R., Alonso, A., Heiss, G., Coresh, J., Mosley, T., Gottesman, R. F., Ballantyne, C. M., & Power, M. C. (2019). ApoB, small-dense LDL-C, Lp(a), LpPLA2 activity, and cognitive change. Neurology, 92(22), e2580–e2593. https://doi.org/10.1212/WNL.0000000000007574
- Premont, R. T., Reynolds, J. D., Zhang, R., & Stamler, J. S. (2020). Role of nitric oxide carried by hemoglobin in cardiovascular physiology. Circulation Research, 126(1), 129–158. https://doi.org/10.1161/circresaha.119.315626
- Pyun, J.-M., Ryoo, N., Park, Y. H., & Kim, S. (2020). Fibrinogen levels and cognitive profile differences in patients with mild cognitive impairment. Dementia and Geriatric Cognitive Disorders, 49(5), 489–496. https://doi.org/10.1159/000510420
- Qi, M., Billheimer, J., Chang, C.-C. H., Janssen, I., Brooks, M. M., Orchard, T., Karlamangla, A. S., Barinas-Mitchell, E., Derby, C. A., McConnell, D., Crawford, S., & Samar. (2024). High-density lipoprotein over midlife and future cognition in women: The SWAN HDL Ancillary Study. The Journal of Clinical Endocrinology & Metabolism, 110(7), 1980–1988. https://doi.org/10.1210/clinem/dgae697
- Rajeev, V., Chai, Y. L., Poh, L., Selvaraji, S., Fann, D. Y., Jo, D.-G., De, M., Drummond, G. R., Sobey, C. G., Arumugam, T. V., Chen, C., & Lai, M. K. P. (2023). Chronic cerebral hypoperfusion: A critical feature in unravelling the etiology of vascular cognitive impairment. Acta Neuropathologica Communications, 11, 93. https://doi.org/10.1186/s40478-023-01590-1
- Razavi, A. C., Mehta, A., Jain, V., Patel, P., Liu, C., Patel, N., Eisenberg, S., Vaccarino, V., Isiadinso, I., Sperling, L. S., & Quyyumi, A. A. (2023). High-density lipoprotein cholesterol in atherosclerotic cardiovascular disease risk assessment: Exploring and explaining the “U”-shaped curve. Current Cardiology Reports, 25(12), 1725–1733. https://doi.org/10.1007/s11886-023-01987-3
- Ridker, P. M., & Cook, N. (2004). Clinical usefulness of very high and very low levels of C-reactive protein across the full range of Framingham Risk Scores. Circulation, 109(16), 1955–1959. https://doi.org/10.1161/01.cir.0000125690.80303.a8
- Rivera Antonio, A. M., Padilla Martínez, I. I., Torres-Ramos, M. A., & Rosales-Hernández, M. C. (2025). Myeloperoxidase as a therapeutic target for oxidative damage in Alzheimer’s disease. Journal of Enzyme Inhibition and Medicinal Chemistry, 40(1). https://doi.org/10.1080/14756366.2025.2456282
- Röhr, F., Bucholtz, N., Toepfer, S., Norman, K., Spira, D., Steinhagen-Thiessen, E., Lill, C. M., Bertram, L., Demuth, I., Buchmann, N., & Düzel, S. (2020). Relationship between lipoprotein (a) and cognitive function – Results from the Berlin Aging Study II. Scientific Reports, 10, 10636. https://doi.org/10.1038/s41598-020-66783-3
- Sachdev, P. S., Bentvelzen, A. C., Gustafson, D., Hansra, G. K., Hosoki, S., Jiang, J., Lennon, M. J., Moro, M. A., Saks, D. G., Samaras, K., Kovacic, J. C., & Kalaria, R. (2026). Vascular cognitive impairment and dementia: Clinical features, neuropathology, and biomarkers. Journal of the American College of Cardiology, 87(1), 52–76. https://doi.org/10.1016/j.jacc.2025.11.008
- Sampson, M., Wolska, A., Warnick, R., Lucero, D., & Remaley, A. T. (2021). A new equation based on the standard lipid panel for calculating small dense low-density lipoprotein-cholesterol and its use as a risk-enhancer test. Clinical Chemistry, 67(7), 987–997. https://doi.org/10.1093/clinchem/hvab048
- Sartori, A. C., Vance, D. E., Slater, L. Z., & Crowe, M. (2012). The impact of inflammation on cognitive function in older adults: Implications for health care practice and research. The Journal of Neuroscience Nursing, 44(4), 206–217. https://doi.org/10.1097/JNN.0b013e3182527690
- Schilling, S., Tzourio, C., Soumaré, A., Kaffashian, S., Dartigues, J.-F., Ancelin, M.-L., Samieri, C., Dufouil, C., & Debette, S. (2017). Differential associations of plasma lipids with incident dementia and dementia subtypes in the 3C Study: A longitudinal, population-based prospective cohort study. PLoS Medicine, 14(3), e1002265. https://doi.org/10.1371/journal.pmed.1002265
- Serrano, J. C. E., Castro-Boqué, E., García-Carrasco, A., Morán-Valero, M. I., González-Hedström, D., Bermúdez-López, M., Valdivielso, J. M., Espinel, A. E., & Portero-Otín, M. (2023). Antihypertensive effects of an optimized aged garlic extract in subjects with grade I hypertension and antihypertensive drug therapy: A randomized, triple-blind controlled trial. Nutrients, 15(17), 3691. https://doi.org/10.3390/nu15173691
- Shimada, Y., Terasawa, M., Okazaki, F., Nakayama, H., Zang, L., Nishiura, K., Matsuda, K., & Nishimura, N. (2021). Rhamnan sulphate from green algae Monostroma nitidum improves constipation with gut microbiome alteration in double-blind placebo-controlled trial. Scientific Reports, 11(1), 13384. https://doi.org/10.1038/s41598-021-92459-7
- Silva, D. V. T. da, Baião, D. dos S., Ferreira, V. F., & Paschoalin, V. M. F. (2022). Betanin as a multipath oxidative stress and inflammation modulator: A beetroot pigment with protective effects on cardiovascular disease pathogenesis. Critical Reviews in Food Science and Nutrition, 62(2), 539–554. https://doi.org/10.1080/10408398.2020.1822277
- Song, F., Poljak, A., Crawford, J. D., Kochan, N. A., Wang, W., Cameron, B., Lux, O., Brodaty, H., Mather, K. A., Smythe, G. A., & Sachdev, P. S. (2012). Plasma apolipoprotein levels are associated with cognitive status and decline in a community cohort of older individuals. PLoS ONE, 7(6), e34078. https://doi.org/10.1371/journal.pone.0034078
- Speer, H., D’Cunha, N. M., Alexopoulos, N. I., McKune, A. J., & Naumovski, N. (2020). Anthocyanins and human health—A focus on oxidative stress, inflammation and disease. Antioxidants (Basel), 9(5), 366. https://doi.org/10.3390/antiox9050366
- State Key Laboratory of Bioengineering, Institute of Process Engineering, & Chinese Academy of Sciences. (2023). Rhamnan sulfate in a proprietary extract of Monostroma nitidum regenerates a compromised endothelial glycocalyx shed caused by high glucose. https://7072026.fs1.hubspotusercontent-na1.net/hubfs/7072026/2023%20Mar%20Rhamnan%20sulfate%20from%20Monostroma%20nitidum%20EGX%20Microfluidic%20Chip%20Study.pdf
- Ştefănescu, C., Voştinaru, O., Mogoşan, C., Crişan, G., & Balica, G. (2025). The neuroprotective potential of betalains: A focused review. Plants (Basel), 14(7), 994. https://doi.org/10.3390/plants14070994
- Suzuki, K., & Terasawa, M. (2020). Biological activities of rhamnan sulfate extract from the green algae Monostroma nitidum (Hitoegusa). Marine Drugs, 18(4), 228. https://doi.org/10.3390/md18040228
- Tsikas, D., Bollenbach, A., Hanff, E., & Kayacelebi, A. A. (2018). Asymmetric dimethylarginine (ADMA), symmetric dimethylarginine (SDMA) and homoarginine (hArg): The ADMA, SDMA and hArg paradoxes. Cardiovascular Diabetology, 17(1), 1. https://doi.org/10.1186/s12933-017-0656-x
- Tzikas, S., Schlak, D., Sopova, K., Gatsiou, A., Stakos, D., Stamatelopoulos, K., Stellos, K., & Laske, C. (2014). Increased myeloperoxidase plasma levels in patients with Alzheimer’s disease. Journal of Alzheimer’s Disease, 39(3), 557–564. https://doi.org/10.3233/jad-131469
- Vaccaro, M. G., Innocenti, B., Cione, E., Gallelli, L., De Sarro, G., Bonilla, D. A., & Cannataro, R. (2023). Acute effects of a chewable beetroot-based supplement on cognitive performance: A double-blind randomized placebo-controlled crossover clinical trial. European Journal of Nutrition, 63(1), 303–321. https://doi.org/10.1007/s00394-023-03265-y
- van Oijen, M., van der Meer, I. M., Hofman, A., Witteman, J. C., Koudstaal, P. J., & Breteler, M. (2006). Lipoprotein‐associated phospholipase A2 is associated with risk of dementia. Annals of Neurology, 59(1), 139–144. https://doi.org/10.1002/ana.20721
- Villaño, D., Marhuenda, J., Arcusa, R., Moreno-Rojas, J. M., Cerdá, B., Pereira-Caro, G., & Zafrilla, P. (2023). Effect of black garlic consumption on endothelial function and lipid profile: A before-and-after study in hypercholesterolemic and non-hypercholesterolemic subjects. Nutrients, 15(14), 3138. https://doi.org/10.3390/nu15143138
- Walldius, G., de Faire, U., Alfredsson, L., Leander, K., Westerholm, P., Malmström, H., Ivert, T., & Hammar, N. (2021). Long-term risk of a major cardiovascular event by apoB, apoA-1, and the apoB/apoA-1 ratio—Experience from the Swedish AMORIS cohort: A cohort study. PLoS Medicine, 18(12), e1003853. https://doi.org/10.1371/journal.pmed.1003853
- Wang, A., Liu, J., Meng, X., Li, J., Wang, H., Wang, Y., Su, Z., Zhang, N., Dai, L., Wang, Y., & Wang, Y. (2018). Association between oxidized low-density lipoprotein and cognitive impairment in patients with ischemic stroke. European Journal of Neurology, 25(1), 185–191. https://doi.org/10.1111/ene.13497
- Ward, M. A., Bendlin, B. B., McLaren, D. G., Hess, T. M., Gallagher, C. L., Kastman, E. K., Rowley, H. A., Asthana, S., Carlsson, C. M., Sager, M. A., & Johnson, S. C. (2010). Low HDL cholesterol is associated with lower gray matter volume in cognitively healthy adults. Frontiers in Aging Neuroscience, 2, 29. https://doi.org/10.3389/fnagi.2010.00029
- Watson, A. W., Scheepens, A., Kennedy, D. O., & Haskell-Ramsay, C. F. (2026). Effects of blackcurrant juice on pre-frontal cortical haemodynamics and cognition in healthy young adults. Nutritional Neuroscience, 29(1), 14–28. https://doi.org/10.1080/1028415x.2025.2538062
- Wingo, A. P., Vattathil, S. M., Liu, J., Fan, W., Cutler, D. J., Levey, A. I., Schneider, J. A., Bennett, D. A., & Wingo, T. S. (2022). LDL cholesterol is associated with higher AD neuropathology burden independent of APOE. Journal of Neurology, Neurosurgery & Psychiatry, 93(9), 930–938. https://doi.org/10.1136/jnnp-2021-328164
- Wright, K. P. Jr, Drake, A. L., Frey, D. J., Fleshner, M., Desouza, C. A., Gronfier, C., & Czeisler, C. A. (2015). Influence of sleep deprivation and circadian misalignment on cortisol, inflammatory markers, and cytokine balance. Brain, Behavior, and Immunity, 47, 24–34. https://doi.org/10.1016/j.bbi.2015.01.004
- Xu, L., Tian, Z., Chen, H., Zhao, Y., & Yang, Y. (2021). Anthocyanins, anthocyanin-rich berries, and cardiovascular risks: Systematic review and meta-analysis of 44 randomized controlled trials and 15 prospective cohort studies. Frontiers in Nutrition, 8, 47884. https://doi.org/10.3389/fnut.2021.747884
- Yates, K. F., Sweat, V., Yau, P. L., Turchiano, M. M., & Convit, A. (2012). Impact of metabolic syndrome on cognition and brain: A selected review of the literature. Arteriosclerosis, Thrombosis, and Vascular Biology, 32(9), 2060–2067. https://doi.org/10.1161/ATVBAHA.112.252759
- Zhao, Y., Zhou, R., Wei, S., Hu, N., Gao, L., Dang, L., Wang, J., Wang, J., Qu, Q., & Shang, S. (2025). The relationship between plasma oxidized low-density lipoprotein and all-cause cognitive impairment: A population-based cross-sectional study in a rural area of Xi’an, China. Journal of Alzheimer’s Disease, 13872877251393712. https://doi.org/10.1177/13872877251393712
- Zhu, Y., Ling, W., Guo, H., Song, F., Ye, Q., Zou, T., Li, D., Zhang, Y., Li, G., Xiao, Y., Liu, F., Li, Z., Shi, Z., & Yang, Y. (2013). Anti-inflammatory effect of purified dietary anthocyanin in adults with hypercholesterolemia: A randomized controlled trial. Nutrition, Metabolism, and Cardiovascular Diseases, 23(9), 843–849. https://doi.org/10.1016/j.numecd.2012.06.005
- Zinellu, A., Tommasi, S., Sedda, S., & Mangoni, A. A. (2023). Circulating arginine metabolites in Alzheimer’s disease and vascular dementia: A systematic review and meta-analysis. Ageing Research Reviews, 92, 102139. https://doi.org/10.1016/j.arr.2023.102139
- Zuo, Q., Song, L., Gao, X., Cen, M., Fu, X., Qin, S., & Wu, J. (2024). Associations of metabolic syndrome with cognitive function and dementia risk: Evidence from the UK Biobank cohort. Diabetes, Obesity & Metabolism, 26(12), 6023–6033. https://doi.org/10.1111/dom.15977
